
In rectal spacing,
control matters.
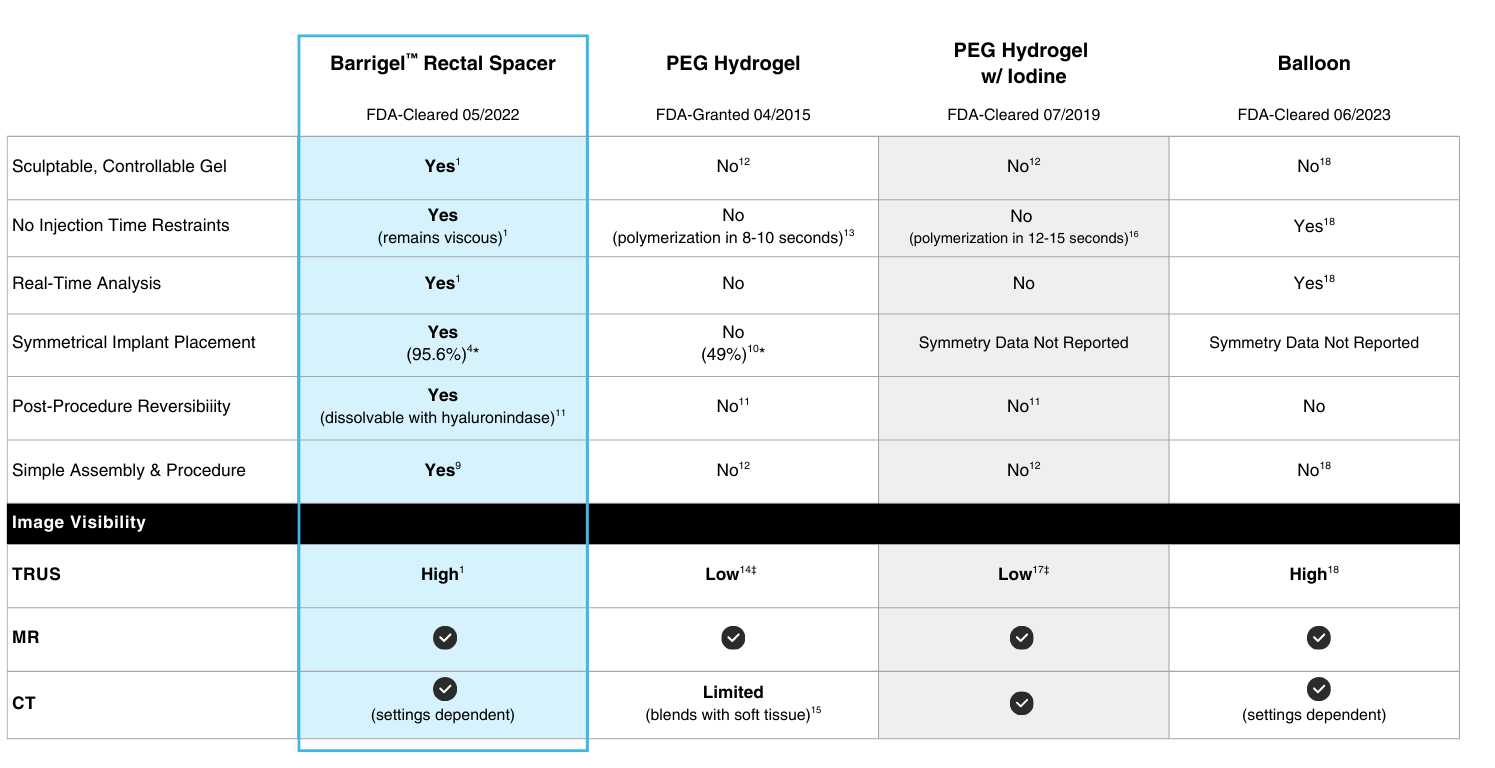
Barrigel™ rectal spacer is the first and only sculptable rectal spacer1-3— allowing you to create space personalized to your patient’s unique anatomy.1,4-6
Rectal spacers reduce GI side effects associated with radiation therapy.
Barrigel™ rectal spacer is proven superior in the reduction of acute grade 2+ GI toxicity compared to control, leading to fewer side effects.1
Published Acute G2+ Toxicity Rate:
No Spacer8-11
Depending on fractionation scheme
10-38%
Acute G2+ Toxicity Rate:
Barrigel™ RCT1
Hypofractionated regimen: 60 Gy in 20 Fractions (3 Gy/Fx)
Control cohort
13.8%
Barrigel™spacer cohort
2.9%
98.5%
OF PATIENTS
Met the primary endpoint of achieving at least a 25% reduction in rectal V54 Gy* (p<0.001) in the Barrigel pivotal trial.1
Acute toxicities have been shown to predict a higher rate of late toxicities12-13
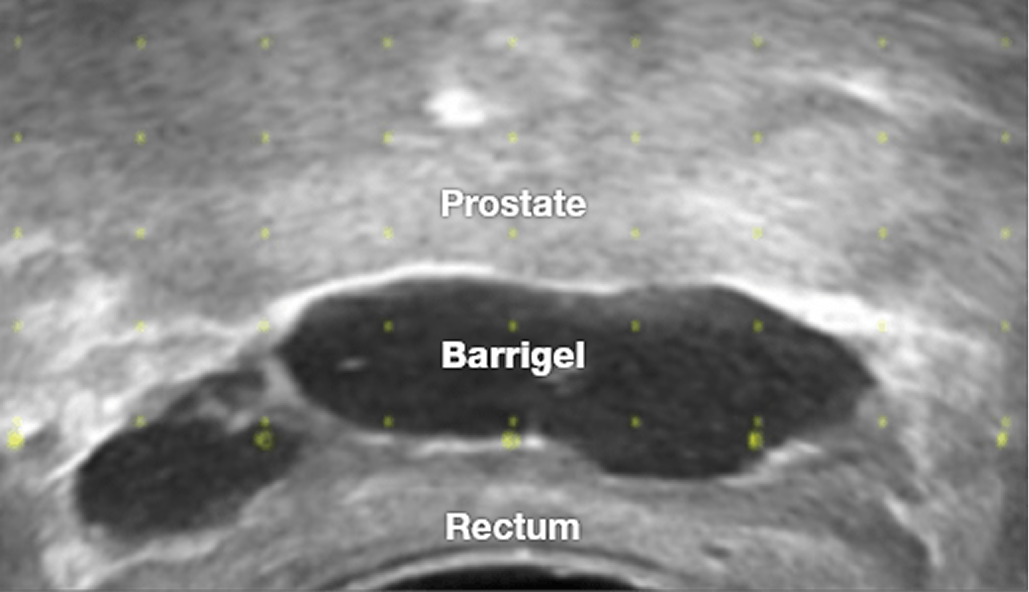
With Barrigel™ rectal spacer, YOU are in control.
First and only sculptable rectal spacer for control over implant shape and placement1-6
No injection time constraints. Non-polymerizing material allows you to sculpt, layer, and adjust in real-time.1,4-6
Highly visible on TRUS. Image-guided procedure with real-time placement visualization.1,4-6
Achieve apical spacing for better bowel quality of life.2
Simple assembly and procedure.7
Take the first step integrating Barrigel™ rectal spacer into your practice. The Barrigel™ Training Program includes a mix of online and live case training, with Barrigel™ Procedural Specialists and physician peers providing support along the way.
Get Started with Barrigel™ Rectal Spacer Training
Hear From Barrigel™ - Trained Physicians
“The ease-of-use injecting Barrigel™ rectal spacer allows the implanter to inject the spacer under no time constraint, and switch easily from sagittal to transverse ultrasound views for symmetrical, excellent coverage of the entire prostate gland. IT'S A NO BRAINER. ”

Zachary Klaassen, MD, MSc**
Urologic Oncologist at Georgia Cancer Center
“My practice began using Barrigel™ spacer because it allows me to take the time necessary to create a symmetric implant that allows for protection of the entire rectum. Whether my patient is receiving IMRT, SBRT, or brachytherapy, Barrigel™ spacer allows me to PROTECT HEALTHY TISSUE from unwanted radiation exposure and reduce side effects.”

Suraj Singh, MD**
Radiation Oncologist Director at Parkview Comprehensive Cancer Center
Control matters for improved symmetry.
BARRIGEL
Sculptable control over implant placement results in more symmetric coverage over the whole posterior rectal/prostate interface.
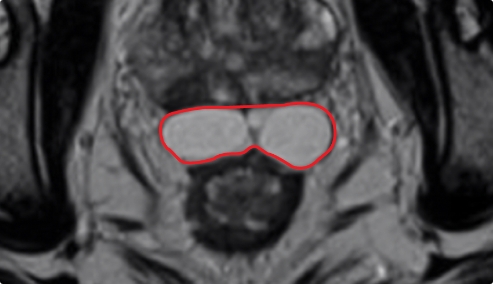
Implant Symmetry RESULTS6
>95%
Pivotal Trial Patients; % of implants centered on prostate midline
PEG HYDROGEL
Lack of control over placement of the implant can result uneven and inconsistent coverage.
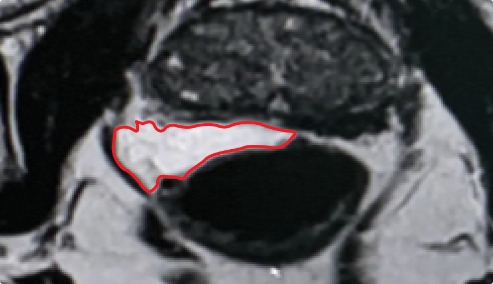
Implant Symmetry RESULTS7
49%
Pivotal Trial Patients; % of implants centered on prostate midline
Rectal spacer properties.
Information provided for educational purposes only. No head-to-head study has been performed. Results from different clinical studies are not directly comparable.
Hear from Barrigel Physicians
“In my experience, Barrigel is safer due to the ability to control its location and alter its placement during the procedure. This allows us to have a MORE UNIFORM RESULT in tougher cases where another procedure could result in delay of care.”

Varun Sundaram, MD
Urologist at Urology Austin in Austin, TX, and Designated Barrigel Center of Excellence
“The ease-of-use injecting Barrigel allows the implanter to inject the spacer under no time constraint, and switch easily from sagittal to transverse ultrasound views for symmetrical, excellent coverage of the entire prostate gland. IT'S A NO BRAINER. ”

Zachary Klaassen, MD, MSc
Urologic Oncologist at Georgia Cancer Center
Resources for Physicians and Staff
Explore clinical articles, educational videos, downloadable resources, event information and more.
Contact us today for more information about Barrigel™ Rectal Spacer
1. Mariados NF, Orio PF III, King MT et al. JAMA Oncol (2023).*
2. King MT, Svatos M, Orio PF III et al. Pract Radiat Oncol (2023).*
3. Data on file. As of 10/01/2024.
4. Svatos M, Chell E, Low DA et al. Med Phys (2024)*
5. Williams J, Mc Millan K, Chao M et al. J Med Imag Radiat Sci (2022).
6. Gejerman G, Goldstein MM, Chao M et al. Pract Radiat Oncol (2023).**
7. Barrigel Injectable Gel Instructions for Use.
8. Fransson P, Nilsson P, Gunnlaugsson A et al. Lancet Oncol (2021).
9. Rodda S, Tyldesley S, Morris JW et al. J Radiat Oncol Biol Phys (2017).
10. Kerkmeijer LGW, Groen VH, Pos FJ et al. J Clin Oncol (2021).
11. Dearnaley D, Syndikus I, Mossop H et al. Lancet Oncol (2016).
12. Michalski JM et al. Int J Radiat Oncol Biol Phys (2010).
13. Vargas C et al. Int J Radiat Oncol Biol Phys (2005).
*Study sponsored by Palette Life Sciences, now part of Teleflex.
**Drs Klaassen, Singh, Gejerman, Chao, Lederer, and Orio are paid consultants of Palette Life Sciences, now part of Teleflex.
Barrigel™ rectal spacer is intended to temporarily position the anterior rectal wall away from the prostate during radiotherapy for prostate cancer and, in creating this space, it is the intent of Barrigel™ rectal spacer to reduce the radiation dose delivered to the anterior rectum. Barrigel™ rectal spacer is composed of biodegradable material and maintains space for the entire course of prostate radiotherapy treatment and is intended to be absorbed by the patient’s body over time.
Barrigel™ rectal spacer should only be administered by qualified and properly trained physicians with experience in ultrasound guidance and injection techniques in the urogenital/pelvic area.
As with any medical treatment, there are some risks involved with the use of Barrigel™ rectal spacer. Potential complications associated with the use of Barrigel™ rectal spacer include, but are not limited to: pain associated with Barrigel™ rectal spacer injection; needle penetration of the bladder, prostate, rectal wall, rectum, or urethra; injection of Barrigel™ rectal spacer into the bladder, prostate, rectal wall, rectum, urethra, or intravascularly; local inflammatory reactions; infection; urinary retention; rectal mucosal damage, ulcers, necrosis; bleeding; constipation; and rectal urgency. More information on indications, contraindications, warnings and instructions for use can be found in the Instructions For Use at www.barrigel.com. Individual results may vary.
Caution: Federal (USA) law restricts this device to sale by or on the order of a physician.
Barrigel, and the Barrigel logo are trademarks or registered trademarks of Teleflex Incorporated or its affiliates, in the U.S. and/or other countries.

©2025 Teleflex Incorporated. All rights reserved. APM1219A


